Proper inference from Simon's two-stage designs
by Tatsuki Koyama, Heidi Chen and Will GrayIntroduction
There are many software available for constructing a Simon's design. These software usually give sample sizes and critical values for stages 1 and 2, therefore, hypothesis testing is simple to conduct. Analyzing the data beyond hypothesis testing, however, is not straightforward when a Simon's design is used. When the actual stage 2 sample size is different from the one originally planned, data analysis is more complicated. Many previously published clinical trials do not correctly account for such mid-course changes.Notation
Response Rate
-
 Response rate under the null hypothesis
Response rate under the null hypothesis
-
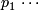 Response rate under the alternative hypothesis
Response rate under the alternative hypothesis
Stage 1
-
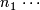 Planned sample size for stage 1.
Planned sample size for stage 1.
-
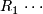 Critical value for stage 1. Note if there are
Critical value for stage 1. Note if there are 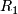 or more ''successes'' in stage 1, the trial continues to stage 2.
or more ''successes'' in stage 1, the trial continues to stage 2.
Stage 2 (planned)
-
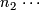 Planned sample size for stage 2.
Planned sample size for stage 2.
-
 Planned critical value at the end of stage 2. Note if there are
Planned critical value at the end of stage 2. Note if there are 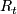 or more ''successes'' in stage 1 and 2 combined, the null hyopthesis is rejected.
or more ''successes'' in stage 1 and 2 combined, the null hyopthesis is rejected.
Stage 2 (actual)
-
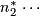 Actual sample size for stage 2.
Actual sample size for stage 2.
Data
-
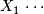 Number of successes in stage 1.
Number of successes in stage 1.
-
 Number of successes in stage 2 (out of
Number of successes in stage 2 (out of 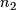 or
or 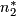 ).
).
 Copyright © 2013-2022 by the contributing authors. All material on this collaboration platform is the property of the contributing authors.
Copyright © 2013-2022 by the contributing authors. All material on this collaboration platform is the property of the contributing authors. Ideas, requests, problems regarding Vanderbilt Biostatistics Wiki? Send feedback