You are here: Vanderbilt Biostatistics Wiki>Main Web>Education>HandoutsBioRes>ClinStat>ThinkSyllabus>STBRsylInference (revision 14)EditAttach
Section 2: Tools for Formal Statistical Inference
Instructor: Tatsuki Koyama
The statistical inferences needed to answer many of the scientific questions addressed in biomedical research take the form of formal hypothesis tests and estimation of effects. For example, a clinical trial can address the question of whether a new therapy is superior to a standard treatment and can provide an assessment of the magnitude of the benefit of the new therapy. This section describes the concepts involved in formal hypothesis testing and estimation, including the interpretation of P-values and confidence intervals.Handouts for 2004.
Learning Objectives
- To illustrate the basic logic behind hypothesis testing using a simple example.
- To discuss the notion of a null and alternative hypothesis.
- To discuss the notion of a P-value and statistical power.
- To demonstrate how sample size can influence statistical power.
- To illustrate the use of a confidence interval.
Required Reading
- K&S Chapters 5-9, Sections 30.2, 33.1, 33.2, 35.1-35.4, 35.6, 35.7
Topics and Key Words
- normal distribution
- central limit theorem
- large sample / small sample
- confidence interval
- hypothesis test
- type I error, type II error, power,
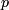 value
value
- type I error, type II error, power,
- paired test
- z test, t test, ANOVA
- fixed effects / random effects
- nonparametric methods
- Bayesian statistics
- sample size calculation
Case Studies and Additional Required Reading
- Sung JJ, Chung SC, Lai CW, Chan FK, Leung JW, Yung My, Kassianides C, Li AK, "Octreotide infusion or emergency sclerotherapy for variceal haemorrhage," Lancet , 342 : 637-41, 1993. pdf here
- Newnham JP, Evans SF, Michael CA, Stanley FJ, Landau LI, "Effects of frequent ultrasound during pregnancy: a randomised controlled trial," Lancet, 342: 887-81, 1993. pdf here
Edit | Attach | Print version | History: r14 < r13 < r12 < r11 | Backlinks | View wiki text | Edit wiki text | More topic actions
Topic revision: r14 - 15 Dec 2009, TheresaScott
 Copyright © 2013-2022 by the contributing authors. All material on this collaboration platform is the property of the contributing authors.
Copyright © 2013-2022 by the contributing authors. All material on this collaboration platform is the property of the contributing authors. Ideas, requests, problems regarding Vanderbilt Biostatistics Wiki? Send feedback
